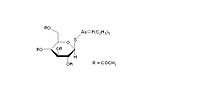
金诺芬
| A+医学百科 >> 金诺芬 |
通用名称:金诺芬
英文名称:Auranofin
英文别名:Aurothioglycolanilide
本品性质稳定,含金约29%,微溶于水,易溶于类脂体中。
目录 |
药理及应用
有抗炎作用,起效慢。口服后所含金的25%被吸收,其中60%与血浆蛋白结合,40%与细胞结合。本品主要由粪便排出。长期服用本品,金血浆浓度12周达高峰,并可保持稳定状态。主用于活动性类风湿性关节炎,亦用于对非甾体抗炎药效果不显或无法耐受患者,可延缓类风湿性关节炎病变发展,改善症状,耐受好。
用法
口服:成人量1日mg,于早饭后服,或早、晚饭后各服3mg。初始剂量1日mg,2周后增至 1日/6mg。如服6个月后疗效不显,可增至一日mg,分3次服。若此量连服3个月效果仍不显,应停药。
注意
(1)常见副作用有腹泻,腹痛、恶心、胃肠不适。尚可见皮疹、瘙痒。口腔炎、结膜炎少见。国外报道有白细胞、血小板减少、紫癜、单纯红细胞发育不全、暂时性蛋白尿、血尿等及肝功能短时异常。
(2)对金过敏、坏死性小肠结肠炎、肺纤维化、剥脱性皮炎、骨髓再生障碍、进行性肾炎、严重肝病患者以及孕妇、哺乳妇女慎用。
[制剂] 片剂:每片3mg。胶囊剂:每胶囊3mg。薄膜片:每片3mg(含金0.87mg)。
药理作用
Classification: Antiarthritic,oral gold compound
Action/Kinetics: Auranofin is agold-containing (29%) compound for PO administration. It has fewer side effects thaninjectable gold products. Although the mechanism is not known, auranofin will improvesymptoms of rheumatoid arthritis; it is most effective in the early stages of activesynovitis and may act by inhibiting sulfhydryl systems. Other possible mechanisms includeinhibition of phagocytic activity of macrophages and polymorphonuclear leukocytes,alteration of biosynthesis of collagen, and alteration of the immune response. Gold willnot reverse damage to joints caused by disease. Approximately 25% of an oral dose isabsorbed. Plasma t1/2 of auranofin gold: 26 days. Onset: 3-4 months(up to 6 months in certain clients). Approximately 3 months are required for steady-stateblood levels to be achieved. The drug is metabolized and excreted in both the urine andfeces.
适应症
Adults and children withrheumatoid arthritis that have not responded to other drugs. Up to 6 months may berequired for beneficial effects to occur. Auranofin should be part of a total treatmentregimen for rheumatoid arthritis, including nondrug treatments.
用法用量
Capsules Rheumatoid arthritis. Adults, initial: Either 6 mg/day or 3 mg b.i.d. If response is unsatisfactory after6 months, increase to 3 mg t.i.d. If response is still inadequate after 3 additionalmonths, discontinue the drug. Dosages greater than 9 mg/day are not recommended. Children, initial: 0.1 mg/kg/day; maintenance: 0.15 mg/kg/day, not to exceed0.2 mg/kg/day. Transfer from injectable gold. Discontinue injectable gold and begin auranofin at a dose of 6 mg/day.
注意事项
Pregnancy Category: C
Contraindications: History ofgold-induced disorders including necrotizing enterocolitis, pulmonary fibrosis,exfoliative dermatitis, bone marrow aplasia, or other hematologic severe disorders. Useduring lactation.
Special Concerns: Use withextreme caution in renal or hepatic disease, skin rashes, marked hypertension, compromisedcerebral or CV circulation, or history of bone marrow depression (e.g., agranulocytopenia,anemia). Gold dermatitis may be aggravated by exposure to sunlight. Although used inchildren, a recommended dosage has not been established. Tolerance to gold is oftendecreased in geriatric clients.
不良反应
GI: N&V, diarrhea (common), abdominal pain, metallic taste, stomatitis, glossitis,gingivitis, anorexia, constipation, flatulence, dyspepsia, dysgeusia, melena. Rarely,dysphagia, GI bleeding, ulcerative enterocolitis. Dermatologic: Skin rashes, pruritus, alopecia, urticaria, angioedema, actinic rash. Hematologic: Leukopenia, anemia, thrombocytopenia (with or without purpura), neutropenia,agranulocytosis, eosinophilia, pancytopenia, hypoplastic anemia, aplasticanemia pure red cell aplasia. Renal: Proteinuria, hematuria. Hepatic: Jaundice (with or without cholestasis, hepatitis with jaundice, toxichepatitis intrahepatic cholestasis. Other: Conjunctivitis,cholestatic jaundice, fever, interstitial pneumonia and fibrosis, peripheral neuropathy.
药物过量
Symptoms: Rapid appearance of hematuria, proteinuria, thrombocytopenia, granulocytopenia. Also,N&V, diarrhea, fever, urticaria, papulovesicular lesions, urticaria, exfoliativedermatitis, pruritus. Treatment: Discontinue promptly and give dimercaprol.Supportive therapy should be provided for renal and hematologic symptoms. Treat moderatelysevere skin and mucous membrane symptoms with topical corticosteroids, oralantihistamines, and anesthetic lotions. Treat severe stomatitis or dermatitis withprednisone, 10-40 mg daily. Treat serious renal, hematologic, pulmonary, and enterocoliticcomplications with prednisone, 40-100 mg daily in divided doses. The duration of treatmentvaries, depending on the severity of symptoms and the response to steroids. In acuteoverdosage, induce emesis or perform gastric lavage immediately.
| 关于“金诺芬”的留言: | |
|
目前暂无留言 | |
| 添加留言 | |